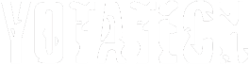"Hydrogen Booster" Setup
#21
For the statistics to work, you need to have the same kind of driving represented both ways. The ideal situation is to have both city and highway randomly mixed.
If you did, say, all city driving for your "before" runs and all highway for your "after" runs, the stats will say there's a difference, but there's no way they can say from what ... the idea is for all the "noise" factors to be randomized on both sides, then they cancel each other out.
If you did, say, all city driving for your "before" runs and all highway for your "after" runs, the stats will say there's a difference, but there's no way they can say from what ... the idea is for all the "noise" factors to be randomized on both sides, then they cancel each other out.
#22
It is quite annoying hearing about grandious fuel mileage claims and the benefits of hybrid engines and hydrogen this and that when I had a car nearly 20 years ago that acheived 48 miles per gallon regularly.
what's changed with cars over the past 20 years that has kept mileage from increasing?
what's changed with cars over the past 20 years that has kept mileage from increasing?
#23
abecedarian, I must disagree with your chemistry. Go look up the molecular weight of Oxygen compared to Hydrogen.
Please do not post on this thread anymore, your comments are no longer along the correct subject of the thread.
Please do not post on this thread anymore, your comments are no longer along the correct subject of the thread.
#24
For the statistics to work, you need to have the same kind of driving represented both ways. The ideal situation is to have both city and highway randomly mixed.
If you did, say, all city driving for your "before" runs and all highway for your "after" runs, the stats will say there's a difference, but there's no way they can say from what ... the idea is for all the "noise" factors to be randomized on both sides, then they cancel each other out.
If you did, say, all city driving for your "before" runs and all highway for your "after" runs, the stats will say there's a difference, but there's no way they can say from what ... the idea is for all the "noise" factors to be randomized on both sides, then they cancel each other out.
you must have apples to apples, so to speak. drive the same city course before and after, and the same highway course before and after... with the same ambient temperature and humidity during both runs of each type.
#25
How should I be measuring your quantities... as molar, avorduopois, electronegativity?
maybe by 'weight' or more appropriately, by 'mass'?
so according to you oxygen is not 1/3 of the byproduct of the electrolysis of H2O, but by chemical/molecular composition... it is.
So?
#29
I am thinking an improved intake would seriously help this mod, I am now looking at what is on the table for cold air intakes. Will also post pictures of this.
Last edited by Wombosi; Jul 7, 2008 at 08:38 PM.
#31
Simple, high-school chemistry teaches you that the energy required to break the bonds of an H2O molecule is more than the energy released by combusting the hydrogen molecules with the oxygen molecules released by the electrolysis reaction.
So I'm at a loss as to how this is supposed to work with 'wasted' energy from your alternator. Perhaps if the hydrogen generating apparatus was powered by a solar source, and could somehow produce hydrogen faster than your engine could consume it, this could become a profitable investment, but until solar cells are more than 50% efficient, I don't see it as a reality... well maybe you could separate the hydrogen at home and pump it into your truck as required, but then again, you're require a significant investment in solar cells or other electrical sources above and beyond your truck engine in order to make the truck into a negative liability.
So I'm at a loss as to how this is supposed to work with 'wasted' energy from your alternator. Perhaps if the hydrogen generating apparatus was powered by a solar source, and could somehow produce hydrogen faster than your engine could consume it, this could become a profitable investment, but until solar cells are more than 50% efficient, I don't see it as a reality... well maybe you could separate the hydrogen at home and pump it into your truck as required, but then again, you're require a significant investment in solar cells or other electrical sources above and beyond your truck engine in order to make the truck into a negative liability.
#32
you can't even 'randomly' mix the statistics for them to be meaningful.
you must have apples to apples, so to speak. drive the same city course before and after, and the same highway course before and after... with the same ambient temperature and humidity during both runs of each type.
you must have apples to apples, so to speak. drive the same city course before and after, and the same highway course before and after... with the same ambient temperature and humidity during both runs of each type.
- You can't be sure that you controlled all the variables,
- That would only show that the system worked/didn't work in the controlled conditions, not necessarily in real life
Therefore, better to have it randomized and have the "noise" on both sides, otherwise, you're just "lying with statistics".
#34
well anyways, as mentioned, maybe in not so simple terms...
I'd be satisfied with controlled testing of an engine on a dynomometer, with it's alternator producing enough hydrogen for the engine to run on.
I'd be satisfied with controlled testing of an engine on a dynomometer, with it's alternator producing enough hydrogen for the engine to run on.
#35
People tend to be passionate about their knowledge and their expertise - let's keep that passion funneled into making this thread more of an asset than a headache.
Thanks,
Fink

#36
#37
well fine, we'll keep the factual information about the energy released by the combustion of hydrogen completely out of the discussion of the energy released from combusting hydrogen in an engine.
good way to make your point.
good way to make your point.
#40
abecedarian I'm starting to think that you believe he is trying to make an electrolysis device to only run his vehicle off hydrogen. If you do believe this, his point is only to produce enough to aid the combustion of the engine.
On another note I don't understand why you are so opposed to the idea of HHO or brown gas. I can understand if you are skeptical as to if it will work or not but there is no need to get so confrontational on the issue. Although I can understand wanting the data of before and after in a certain way. But a dyno will not test if this works in real situations only in ideal situations.
I'm all for trying this idea out, I am a little concerned as to long term effects but that data will come in with time I'm sure. Good for you giving this a try I would if I had the time and the means to.
EDIT: agreed on the point of energy consumption of turning water in to hydrogen gas, this is why any hydrogen produced for the few hydrogen cars on the road today is produced from petroleum
On another note I don't understand why you are so opposed to the idea of HHO or brown gas. I can understand if you are skeptical as to if it will work or not but there is no need to get so confrontational on the issue. Although I can understand wanting the data of before and after in a certain way. But a dyno will not test if this works in real situations only in ideal situations.
I'm all for trying this idea out, I am a little concerned as to long term effects but that data will come in with time I'm sure. Good for you giving this a try I would if I had the time and the means to.
EDIT: agreed on the point of energy consumption of turning water in to hydrogen gas, this is why any hydrogen produced for the few hydrogen cars on the road today is produced from petroleum
Last edited by Red3.slow; Jul 7, 2008 at 09:01 PM.